Review Process
HOW IT WORKS
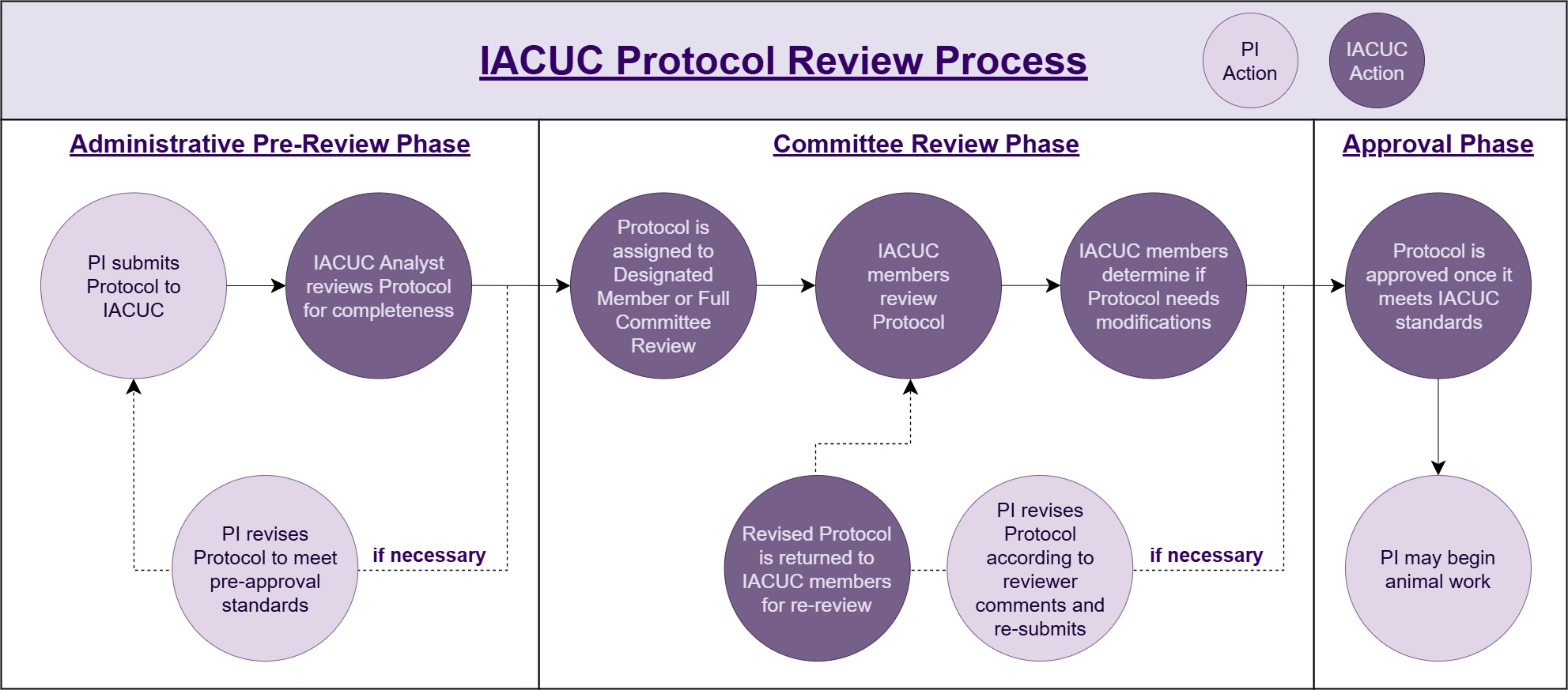
- New protocol submissions are reviewed by either designated member or full committee review (FCR).
- Designated reviews are completed by at least one IACUC member.
- Full committee review (FCR) involves primary and secondary reviewers presenting to the full committee at a convened committee meeting.
Committee members are provided access to all submissions. The Chair/designee appoints 3-4 members as designated reviewers of each submission. Most submissions fall under designated review, which does not have a submission deadline. Some submissions are designated for full committee review (FCR), and require review at a convened meeting of the IACUC. Details for what protocols are automatically sent to FCR can be found in the policy IACUC Review of Animal Study Protocols and Addenda Requests. Protocols meeting the criteria for FCR must be submitted 10 days before a scheduled meeting to meet the deadline to be reviewed at the next meeting. All protocols are fully re-reviewed every 3 years.
APPROVAL
IACUC approval means that animal work approved in the protocol may begin. The PI may order animals from the Center for Comparative Medicine (CCM). Project personnel may gain access to the animal facilities. The PI may talk to Sponsored Research (SR) about opening accounts to spend grant awards.
Details about approval:
- The IACUC approves protocols for a three-year period, even if sponsored funding is for a longer term.
- A de novo (new) protocol must be submitted to the IACUC prior to the end of the initial three-year approval period to avoid protocol inactivation.