The Protocol Review Process
HOW IT WORKS
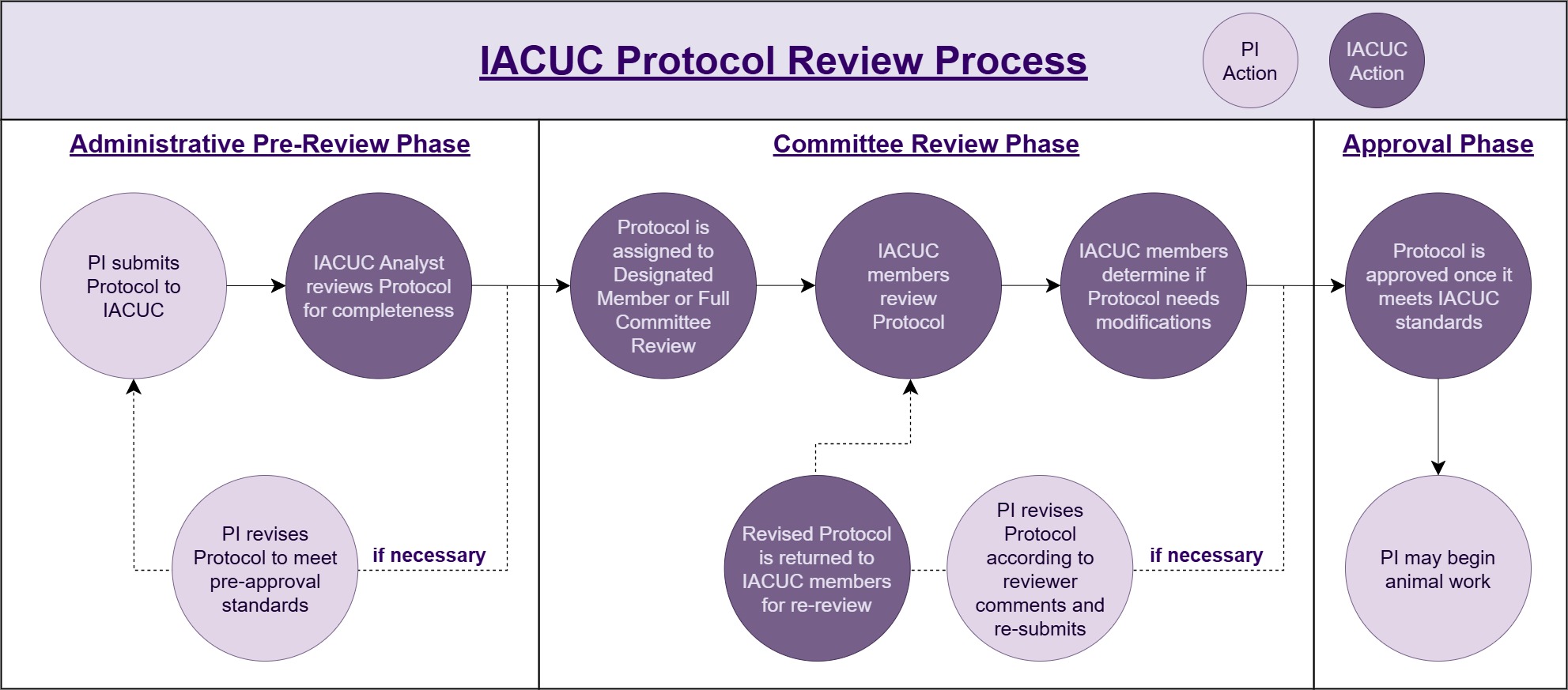
New protocol submissions are reviewed by either designated member or full committee review.
- Designated member reviews (DMR) are completed by at least one voting IACUC member appointed by the Chair/designee. A veterinarian and research safety staff may also be assigned to concurrently review submissions. Most submissions fall under DMR, which does not have a submission deadline.
- Full committee review (FCR) involves review of a submission by the full committee at a convened committee meeting. Details for what protocols are automatically sent to full committee review can be found in the policy on IACUC Review of Animal Study Protocols and Addenda Requests. Protocols meeting the criteria for FCR must be submitted 10 days before a scheduled meeting to meet the deadline to be reviewed during that meeting. See the Meeting Schedule page for submission deadlines.
Protocols discussed but not approved at a full committee meeting may continue through the DMR as per the IACUC Policy – IACUC Process for ASP Review After Full Committee Review (FCR).
Just-In-Time Submission and Review
Following peer review and notification of impact score/percentile, it is strongly recommended that a protocol be submitted for IACUC review. There may be situations when this may not be possible due to changes in how grants are reviewed and funded. The IACUC has implemented a process to address urgent JIT deadlines. Please review the IACUC Chair and IACUC Office memos.
Note that if, during protocol review, it was identified that the protocol must be placed on hold for any reason (e.g., bio-registration/eIBC update, training requirements not met, OHS enrollment not completed), the IACUC review process will continue until all reviewers have motioned for approval on the submission.
If eIBC, training, or OHS enrollment steps are not addressed before the IACUC is ready to approve the submission, a letter will be provided for JIT documentation purposes.
If a protocol requires full committee review, it will be added to the upcoming meeting agenda. It is essential that labs continue to respond to reviewer comments prior to the FCR meeting. If you have any questions, contact the IACUC Office at acuc@northwestern.edu .
APPROVAL
IACUC approval means that animal work approved in the protocol may begin. Once the approval letter is sent in eIACUC, select protocol information is pushed into CCM’s Animal Operations (AOPs). The AOPs protocol will remain in the “awaiting set up” state until a chart string is provided to the CCM Business Office. More information can be found on CCM’s billing page. Once AOPs set up is complete, the PI may order animals from the Center for Comparative Medicine (CCM). Project personnel may gain access to the animal facilities. The PI may talk to Sponsored Research (SR) about opening accounts to spend grant awards.
Details about approval:
- The IACUC approves protocols for a three-year period as required by regulations, even if sponsored funding is for a longer term.
- A De Novo (new) protocol must be submitted to the IACUC prior to the end of the initial three-year approval period to avoid inactivation.